Opinion Piece by: Alexandra Rasnitsyn
You are made from 37.2 trillion cells. Each a small building block that eats, breaths and interacts with other cells to run your body. Cells need to share oxygen and nutrients, deliver complex signals, and control their growth to ensure that all your tissues and organs are in order.
How do these small units co-operate so well?
Our ancestors were “single celled”, or one cell, that looked out only for themselves. These single cell organisms (also known as ‘unicellular organisms’), independent and self-contained, led solitary lives and were invested only in gathering nutrients and making copies of themselves.
Around 1 billion years ago, some unicellular organisms started to cooperate at different points of their life and the first form of multicellularity appeared. In this simple form of co-operation between multiple cells, organisms lived part of their lives as independent single cells and the other as a colony that shared resources. Multicellular life continued to evolve, and around 600 million years ago, the first animals appeared. Animals had a more complicated form of multicellularity, where many cells co-operated throughout their lifetime. In animals, cells formed tissues and organs, each with their unique roles in ensuring the wellbeing of the multicellular organism.
Now, consider your body and the trillions of cells that live together in it, how would multicellular co-operation work on such a large scale?
To ensure we grow and develop correctly and function well as adults, there are several core rules that cells need to obey. Cells need to mature and form tissues in a process known as cell differentiation. Cells also need to control their growth to respect the territory of other cells and ensure that different organs are maintained. Resources like oxygen and nutrients also need to be divided fairly between different tissues.
Curiously, these rules are disrupted in cancer.
Cancer cells betray the rules of co-operation and go back to selfish behaviour. Cancer cells block differentiation and grow uncontrollably and with time, they can invade other tissues in a process known as metastasis. Cancer cells also manipulate and steal resources from their law-abiding, co-operative cell neighbours. For example, cancer cells can deprive non-cancerous cells of oxygen by growing new blood vessels to support themselves.
An emerging theory focuses on the connection between disrupted multicellular co-operation and cancer: Cancer atavism. Atavism, in biology, refers to the reappearance of ancestral traits after being lost during evolution. The cancer atavism theory sees cancer development as an evolutionary throwback, where co-operative cells return to the behaviour of their single cell ancestors. It suggests that when cells undergo mutations that disrupt co-operative behaviour, they become more selfish and gradually transition into a self-absorbed unicellular state. In a unicellular state, cells “cheat” on their multicellular agreement and start to grow uncontrollably and take over our body’s resources.
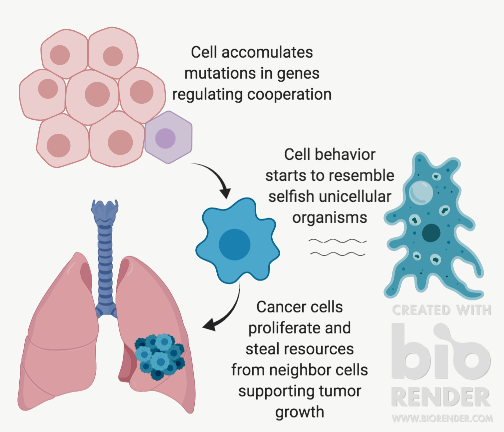
- Cancer Atavism Theory Overview by Alexandra Rasnitsyn, created with BioRender.com
A recent study looked at the evolutionary origins of genes active in tumor cells compared to normal tissue in multiple different cancers. The researchers found that tumours expressed more genes that emerged in early single celled organisms compared to normal tissues. Tumours also deactivated the expression of genes that evolved in animals that are associated with multicellular co-operation, supporting a similarity between cancer and unicellular organisms. In addition, they discovered a network of genes which regulated the transition from multicellular to unicellular states, which might offer new therapeutic directions.
Cancer as an evolutionary throwback is an emerging theory which is gaining support in recent years. Better understanding of the multicellular to unicellular transition in cancer and its evolutionary origins can offer us a new look into how cancer behaves. By exploring the evolutionary mechanisms driving selfish behaviour in cancer cells we may find therapeutic targets which would target cancer cells with ancient unicellular behaviours while leaving normal co-operative cells unharmed.
Alexandra Rasnitsyn is a PhD candidate at the University of Toronto. She is investigating the molecular mechanisms leading to higher risk of childhood brain cancer in boys compared to girls.
References
- Aktipis, C.A., Boddy, A.M., Jansen, G., Hibner, U., Hochberg, M.E., Maley, C.C. and Wilkinson, G.S. 2015. Cancer across the tree of life: cooperation and cheating in multicellularity. Philosophical transactions of the Royal Society of London. Series B, Biological sciences. 370, 1673 (Jul. 2015). DOI: https://doi.org/10.1098/rstb.2014.0219.
- Bussey, K.J., Cisneros, L.H., Lineweaver, C.H. and Davies, P.C.W. 2017. Ancestral gene regulatory networks drive cancer. Proceedings of the National Academy of Sciences of the United States of America. 114, 24 (Jun. 2017), 6160–6162. DOI: https://doi.org/10.1073/pnas.1706990114.
- Casás-Selves, M. and Degregori, J. 2011. How cancer shapes evolution, and how evolution shapes cancer. Evolution. 4, 4 (Dec. 2011), 624–634. DOI: https://doi.org/10.1007/s12052-011-0373-y.
- Davies, P.C.W. and Lineweaver, C.H. 2011. Cancer tumors as Metazoa 1.0: tapping genes of ancient ancestors. Physical biology. 8, 1 (Feb. 2011), 015001. DOI: https://doi.org/10.1088/1478-3975/8/1/015001.
- Hedges, S. and Kumar, S. 2009. The Timetree of Life. Oxford University Press.
- Trigos, A.S., Pearson, R.B., Papenfuss, A.T. and Goode, D.L. 2017. Altered interactions between unicellular and multicellular genes drive hallmarks of transformation in a diverse range of solid tumors. Proceedings of the National Academy of Sciences of the United States of America. 114, 24 (2017), 6406–6411. DOI: https://doi.org/10.1073/pnas.1617743114.
- Trigos, A.S., Pearson, R.B., Papenfuss, A.T. and Goode, D.L. 2018. How the evolution of multicellularity set the stage for cancer. British Journal of Cancer. 118, 2 (Jan. 2018), 145–152. DOI: https://doi.org/10.1038/bjc.2017.398.
